Effect of Buffer on Protein Stability in Aqueous Solutions: A Simple Protein Aggregation Model | The Journal of Physical Chemistry B

Low-Conductivity Buffers for High-Sensitivity NMR Measurements | Journal of the American Chemical Society

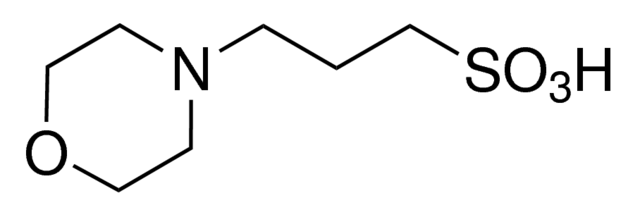
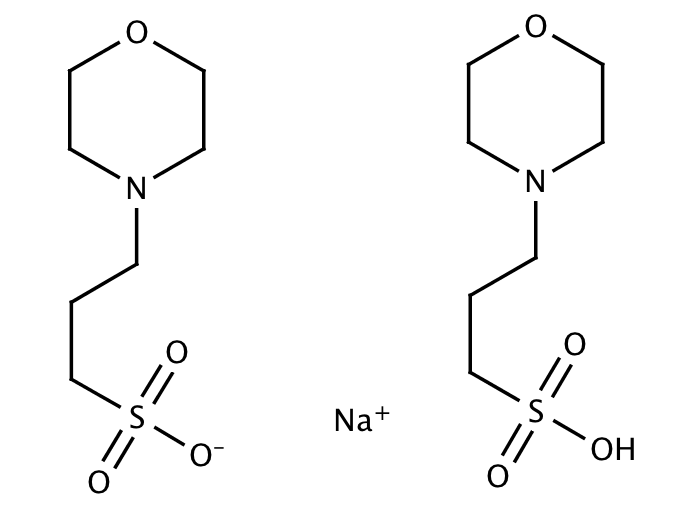
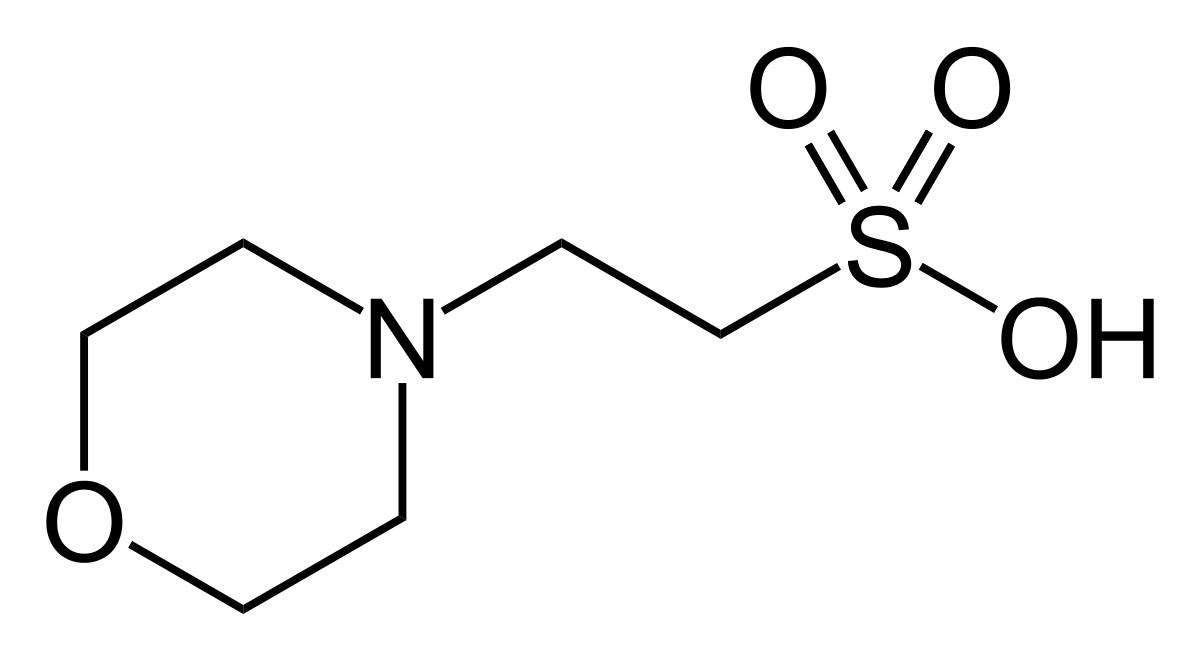
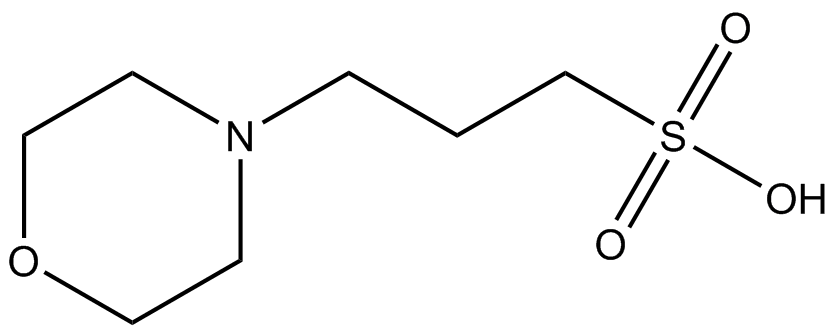
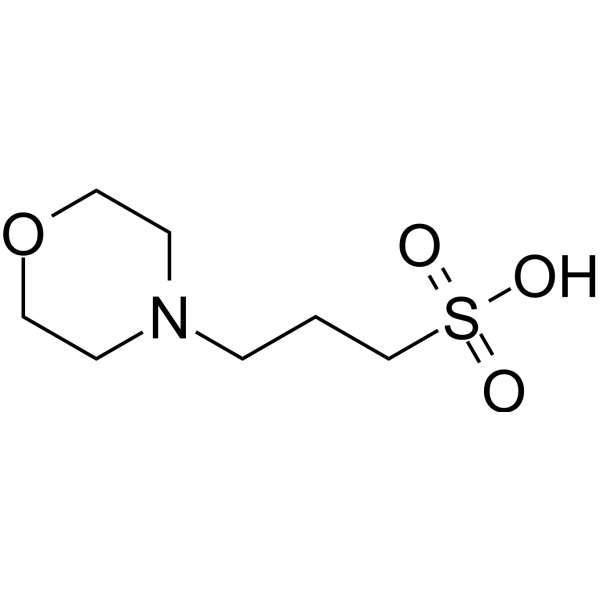
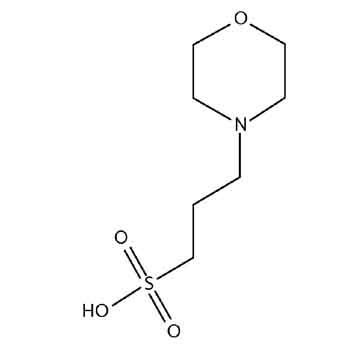
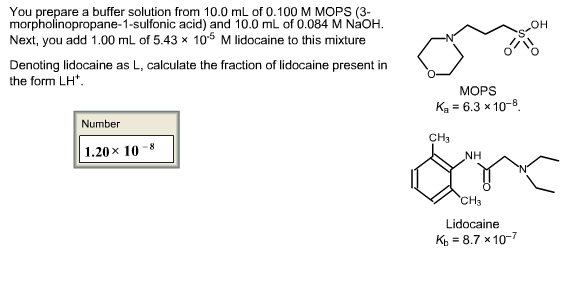
You prepare a buffer solution from 10.0 mL of 0.100 M MOPS (3- morpholinopropane-1-sulfonic acid) and 10.0 mL of 0.077 M NaOH. Next, you add 1.00 mL of 2.05 x 10^-5 M











![MOPS-SDS Running Buffer [20X] - Cepham Life Sciences Research Products MOPS-SDS Running Buffer [20X] - Cepham Life Sciences Research Products](https://www.cephamls.com/wp-content/uploads/2019/02/MOPS-SDS-Running-Buffer-500ml-1-scaled.jpg)

![MOPS Running Buffer [10X] - Cepham Life Sciences Research Products MOPS Running Buffer [10X] - Cepham Life Sciences Research Products](https://www.cephamls.com/wp-content/uploads/2019/02/10379-MOPS_Running_Buffer_1-L-scaled.jpg)